Automated method for large-area imaging of the corneal sub-basal nerve plexus as basis for a reliable biomarker for the assessment of diabetic neuropathy
- Contact:
Dr. Bernd Köhler
- Project Group:
System Integration - Inspection and Digital Image Processing (SI-IIP)
- Funding:
DFG
- Partner:
Rostock University Medical Center
- Startdate:
2016
- Enddate: 2018
Diabetic neuropathy (DN) is the most common long-term complication associated with diabetes mellitus. Diagnosis of DN currently relies on assessments of nerve function that detect pathologic alterations only after occurrence of preliminary clinical symptoms. Objective diagnostic methods with increased sensitivity are urgently needed to facilitate early diagnosis as well as follow-ups and to evaluate novel therapeutic approaches for DN. High expectations are currently placed on confocal laser scanning microscopy of the cornea (CCM). It allows non-invasive in vivo imaging of the nerve fibers constituting the corneal sub-basal nerve plexus (SNP) and enables direct assessment of the condition of small peripheral nerve fibers. Since peripheral neuropathies are observable in the SNP at an early stage, CCM has the potential to be used as a reliable biomarker. A major limitation of this methodology arises from the inhomogeneous distribution of nerve fibers across the area of the cornea. The acquisition of reliable morphological values therefore requires the assessment of a sufficiently large and precisely localized area of the SNP. Moreover, contacting the corneal surface with the microscope causes reversible tissue deformations that disturb the normal arrangement of the SNP parallel to the corneal surface and consequently prevent imaging of the nerve structures across the entire field of view. Finally, the image acquisition process, and frequently the subsequent analysis of the image data as well, is currently characterized almost exclusively by manual tasks and subjective decisions. These limitations of CCM reduce the sensitivity and specificity of the method.
The present project proposes the development of an automated technique capable of generating large-scale images of the SNP for its reliable morphometric characterization as a potential solution. A key innovative aspect of the proposal is an automated control approach to keep the SNP in focus using novel real-time algorithms for tissue classification. The development of novel approaches for minimizing tissue deformations are required in order to reduce image artifacts that currently occur frequently in SNP imaging. Apart from this, another important research goal is the development of new approaches for the precise positioning of the microscope on the corneal surface prior to the imaging process. These are complemented by mathematical methods for verifying and correcting the captured position relative to the corneal apex during recording of the image sequence. The lateral expansion of the imaged area of the SNP is to be achieved by guided eye movements. Finally, the presented work plan includes the development of image processing algorithms to generate mosaic images from the acquired CCM image sequences. These mosaic images enable the robust morphometric characterization of the structure of the SNP and form the basis for a reliable biomarker for the assessment of peripheral neuropathies.
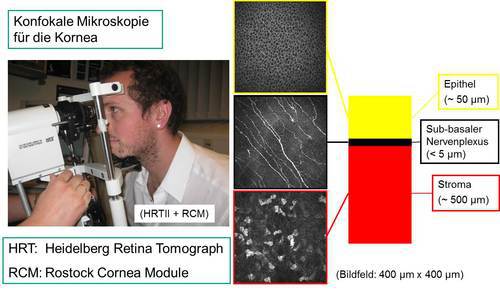
the human cornea

sub-basal nerve plexus (bottom left: single image)
Team:
Dr. Bernd Köhler, Dr. Stephan Allgeier, Prof. Dr. Ralf Mikut
Karlsruhe Institute of Technology (KIT), Institute for Automation and Applied Informatics
Prof. Dr. Oliver Stachs
Rostock University Medical Center, Department of Ophthalmology
Key publications:
Allgeier S, Bartschat A, Bohn S, Peschel S, Reichert KM, Sperlich K, Walckling M, Hagenmeyer V, Mikut R, Stachs O, Köhler B. 3D confocal laser-scanning microscopy for large-area imaging of the corneal subbasal nerve plexus. Sci Rep. 2018;8:7468. doi:10.1038/s41598-018-25915-6.
Bohn S, Sperlich K, Allgeier S, Bartschat A, Prakasam R, Reichert KM, Stolz H, Guthoff R, Mikut R, Köhler B, Stachs O. Cellular in vivo 3D imaging of the cornea by confocal laser scanning microscopy. Biomed Opt Express. 2018;9(6):2511-2525. doi:10.1364/BOE.9.002511
Bartschat A, Stegmaier J, Allgeier S, Reichert KM, Bohn S, Stachs O, Köhler B, Mikut R. Augmentations of the bag of visual words approach for real-time fuzzy and partial image classification. In: Hoffmann F, Hüllermeier E, Mikut R (Hrsg.). Proc. 27. Workshop Computational Intelligence. Karlsruhe: KIT Scientific Publishing; 2017. pp. 227-242. doi:10.5445/KSP/1000074341

